The fourth most common neurological disorder in the world is epilepsy, a disease famously characterized by unpredictable seizures. Be that as it may, the condition exists as more of a spectrum, with variances depending on the afflicted individual’s biological makeup. According to medical experts, a person is diagnosed with epilepsy if they have two, or more, unprovoked seizures not caused a previous condition like low blood sugar.
According to statistics released by Healthline, 1.2% of Americans will be diagnosed with the disease, resulting in about 3.4 million people affected nationwide, and on a global scale, epilepsy affects 1 in 26 people, totaling to over 65 million with the disease. Epilepsy dramatically affects the quality of life for those diagnosed with the disease.
Typically when a patient has had two or more unprovoked seizures and is subsequently diagnosed with epilepsy, they are started on an antiepileptic drug (AED) treatment. According to Dr. J.W. Sander, an expert on clinical and experimental epilepsy, seventy to eighty percent of patients receiving AED treatment “will eventually become seizure free” but the “long-term side effects” of the current AED drugs available often deter patients from opting into treatment.
Additionally, patients who’ve decided to look past the harmful side effects have historically not responded well, overall, to available medications. Pharmaceutical companies have worked endlessly to create a new alternative to traditional epilepsy treatment regimens, and one company, in particular, thought to utilize cannabis in their cocktails.
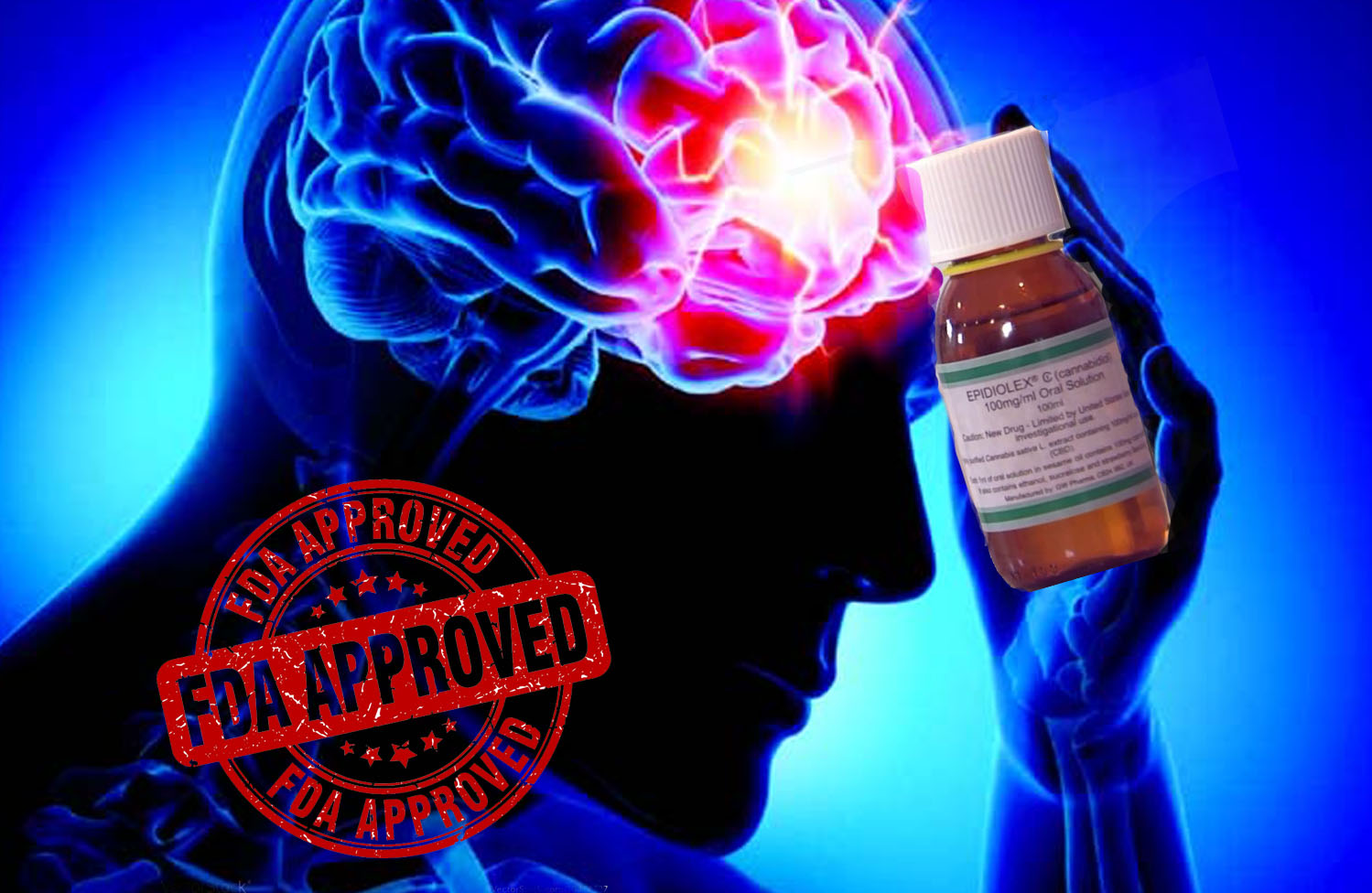
Last year, at the annual meeting of the American Epilepsy Society (AES), GW Pharmaceuticals plc (GWPH) whose work primarily focuses on “discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform,” announced that it had completed Phase 3 trials for a new drug called Epidiolex. The drug, taken in the form of a twice-daily oral solution, was designed to treat patients suffering from Lennox-Gastaut syndrome (LGS), a type of epilepsy that in the early stages of a child’s life, between ages 3 and 5.
GW Pharmaceuticals (GWPH) demonstrated to the attendees of the annual conference that their new drug was incredibly useful for treating patients affected with LGS. According to their findings, of the 23 patients who participated in the 3-month treatment trial, “40% had a >50% reduction in seizures,” indicative of a promising treatment option for the epileptic community and a significant potential investment for investors in the biotech market.
Back in April, Epidiolex was recommended for approval by an advisory committee and submitted to the US Food and Drug Administration. After months of hurdles and challenges endured by GW Pharmaceuticals (GWPH), the drug received approval for distribution in all 50 states on Thursday, according to CNN.
Justin Gover, GW’s (GWPH) chief executive, shared his excitement for Epidiolex’s FDA approval in a written statement on Thursday:
“BECAUSE THESE PATIENTS HAVE HISTORICALLY NOT RESPONDED WELL TO AVAILABLE SEIZURE MEDICATIONS, THERE HAS BEEN A DIRE NEED FOR NEW THERAPIES THAT AIM TO REDUCE THE FREQUENCY AND IMPACT OF SEIZURES. WE ARE COMMITTED TO ENSURING THAT THESE PATIENTS CAN ACCESS THIS NOVEL CANNABINOID MEDICINE THAT HAS BEEN THOROUGHLY STUDIED IN CLINICAL TRIALS, MANUFACTURED TO ASSURE QUALITY AND CONSISTENCY, AND IS ELIGIBLE TO BE COVERED BY INSURANCE FOR APPROPRIATE PATIENTS.”
-JUSTIN GOVER, CHIEF EXECUTIVE OFFICER, GW PHARMACEUTICALS
The FDA’s approval of a cannabinoid-derived drug signifies rising support for marijuana in the pharmaceutical industry as well as in the medical community. In early October, Canadian Prime Minister Justin Trudeau and Parliament legalized cannabis for recreational/medicinal use across the Great White North.
For a plant that has had a history of global prohibition, investors and consumers are fully onboard with the controlled substance. Hopefully, as time goes on, more pharmaceutical companies will follow in GW’s (GWPH) footsteps and use cannabis to treat the many diseases that affect millions around the world.
MAPH Enterprises, LLC | (305) 414-0128 | 1501 Venera Ave, Coral Gables, FL 33146 | new@marijuanastocks.com