The recent events surrounding natural killer (NK) cell therapy have begun to raise some eyebrows and we’re here to discuss a company that may have the right team and treatment to bring NK cell treatment from the clinical stages to the commercial platform. The biotechnology sector has gotten a jolt, especially for early-stage clinical companies.
Affimed (AFMD) entered into a strategic collaboration agreement with Genentech, a member of the Roche Group (RHHBY) and will receive $96 million upfront and committed funding as well as eligibility for up to an additional $5.0 billion including milestone payments, and royalties on sales. The focus: To develop and commercialize novel NK cell engager-based immunotherapeutics to treat multiple cancers.

The new advancements with T-Cell therapies have brought about new hope for a more targeted approach to oncology and the approval by the FDA for drugs like Kymriah from Novartis and Yescarta from Gilead (GILD) have opened the eyes of the pharmaceutical community to what CAR-T treatments, a more targeted form of cancer-fighting therapy can do for patients in all phases of numerous cancers.
But a large downfall has been the overall cost to the patient with some treatments costing nearly $500,000. This is where GT Biopharma (GTBP) could be well positioned to take advantage of both the advancements from T-cell treatment as well as the new attention on NK cell therapy to drive real value not only for investors but also for the entire biopharma community.
GT Biopharma (GTBP) Overview
GT Biopharma Inc. is a clinical-stage biopharmaceutical company that is focused on the development and commercialization of immune-oncology products for patients with hematological malignancies, sarcomas, and solid tumors. GT is also developing a portfolio of products that also prevent motion sickness, muscle weakness for people with myasthenia gravis and lower the effects of nerve pain.
The company’s immuno-oncology portfolio is based off a proprietary technology platform consisting of single-chain bi-, tri- and tetra-specific scFv constructs, combined with proprietary antibody-drug linkers and drug payloads. The central nervous system product portfolio consists of innovative reformulations and/or repurposing of existing therapies that are covered by issued or filed composition of matter patents.
OXS-1550 Near-Term Catalysts
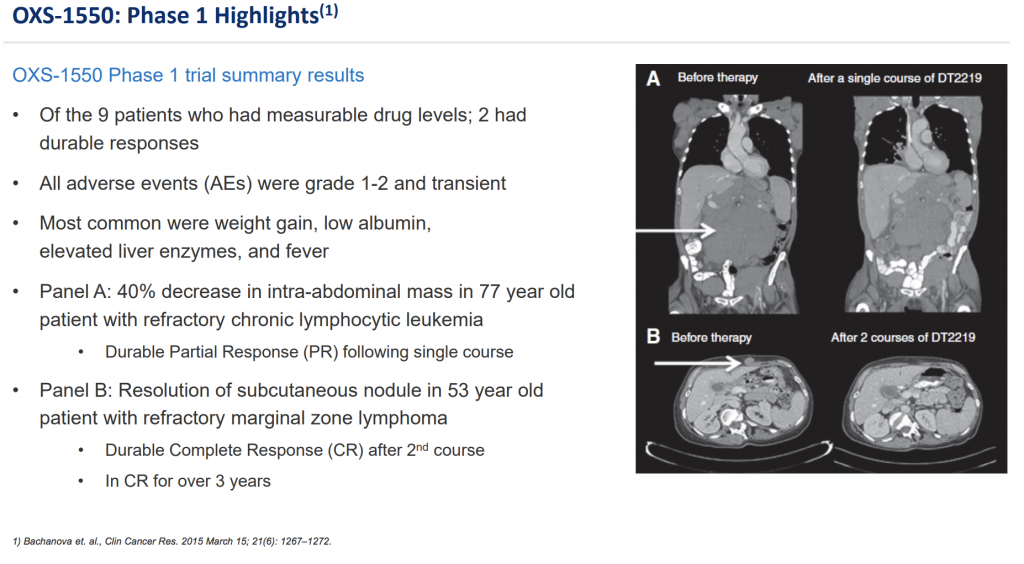
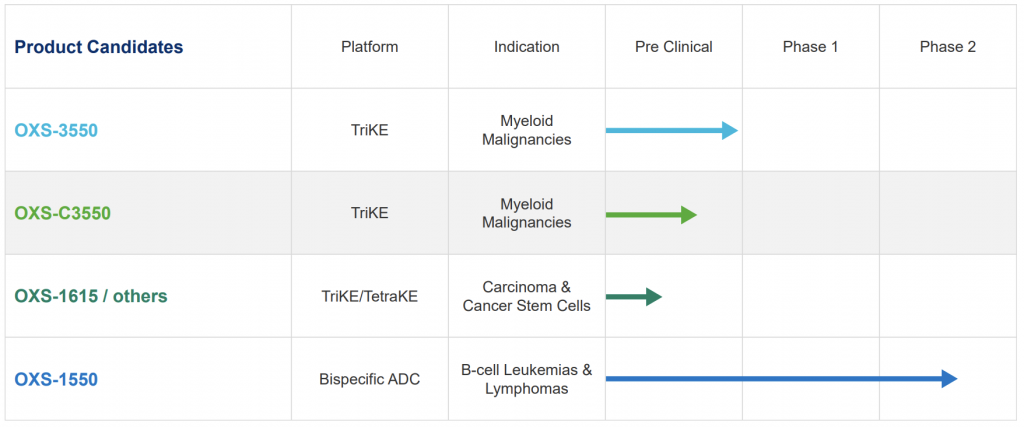
Most recently the company has put a strong focus on its OXS-1550. This targets CD19 & CD22 antigens on select cancer cells with an indication of B-cell lymphomas and leukemias. Not only is OXS-1550 in Phase 2 trials, GT Biopharma will be conducting a combination trial with ibrutinib, a multi-billion dollar drug that has shown immense promise alone and with other combination treatments.
Dr. Daniel Vallera, Director, Section of Molecular Cancer Therapeutics at the Masonic Cancer Center, University of Minnesota who is heading this effort believes combination therapies like these that kill cancer cells based on entirely different mechanisms are the future of cancer treatment.
Known as TriKEs, this type of therapy has the protein version of CAR-T but with multiple clinical and practical advantages according to Dr. Jeffrey Miller, Deputy Director of the Masonic Cancer Center, University of Minnesota. This offers a real opportunity for GT Biopharma (GTBP) to take advantage of the cost benefits for its therapy compared to a more costly CAR-T treatment.
GT Biopharma stated that its platform technologies have the potential to treat large patient populations and substantial markets. The biggest breakthrough could be yet to come. GT Biopharma (GTBP) has additional therapies that are all in different phases of clinical/preclinical development.
Several developments could also pose a near-term opportunity for the company outside of its oncology pipeline:
GTP-004 Progress
GT Biopharma (GTBP) has completed the analysis of pharmacokinetic data from its Phase 1 clinical trial for GTP-004. This therapy is its promising treatment for the symptoms of myasthenia gravis. Based on these and additional data from the Phase 1 clinical trial expects to potentially accelerate the start of its Phase 2 trial in myasthenia gravis patients to the third quarter of 2018.
GTP-011 Progress
GT Biopharma (GTBP) completed primary enrollment in its single-blind, placebo-controlled, cross-over study for its motion sickness drug, GTP-011. GTP-011 is a 72-hour, transdermal formulation that contains a muscarinic receptor antagonist. Unlike scopolamine (Transderm Scop® from Novartis), GTP-011’s active ingredient has been reported not to affect memory and cognition and has a low incidence of sedation (Kay et al., 2012). GTP-011 could be a more favorable alternative.
GT Biopharma Leadership
GT’s management team has a wealth of experience specifically in the clinical and preclinical stage development of biopharmaceutical companies. Dr. Raymond Urbanski holds the position of President and Chief Executive Officer. Dr. Urbanski spent eight years with Pfizer; holding several positions of increasing responsibility with the company, including Vice President/CMO of the Established Products Business Unit, senior medical director of oncology clinical R&D, senior medical director of breast cancer products, and medical director of diversified products.
He has been involved in every phase of drug development and brings extensive experience in developing and overseeing clinical studies, including studies for sunitinib (Sutent), exemestane (Aromasin), irinotecan (Camptosar), epirubicin (Ellence), axitinib, IGF1R inhibitor, and tremelimumab.
In addition to his role with Pfizer, Dr. Urbanski served as Chief Medical Officer of Mylan Inc., Chief Medical Officer of Metabolex Inc., and Senior Director of US Medical Affairs for Aventis.
As the Vice Chairwoman and President Neurology Division, Dr. Kathleen Clarence-Smith brings decades of pipeline development and corporate strategy to GT Biopharma’s team. As the co-founder of Chase Pharmaceuticals, she served as the Chairman of the company’s board for years (2008-2014) and through her time there, she helped build Chase into an organization that was eventually acquired by Allergan, PLC in 2016. In all reality, the deal could be valued at nearly $900 million with milestones.
Furthermore, as the head of CNS development at Sanofi, Hoffmann-La Roche, and Otsuka, Dr. Clarence-Smith developed, secured approval for and supported the launches of significant neurological treatments. This includes Abilify® (aripiprazole), which is a $7 billion peak-year sales pharmaceutical drug used in the treatment of schizophrenia and bipolar disorder.
The company has also amassed a strategic grouping of board & advisory members as well that include the likes of finance executives, corporate structure professionals and biopharmaceutical leaders who can be seen in the company’s most current July 2018 Company Overview.
Potential Value Proposition For Investors
Early, clinical stage companies like GT Biopharma present a number of value propositions for investors. Though many of these companies can hold higher risk due to lack of revenues, incurring high levels of expenses due to research, development, and SG&A, they can also offer higher rewards if early successes are realized.
So far, GT Biopharma has come up favorably with respect to trial results on more than just one of its therapies in the pipeline. Certainly, though the most recent developments with it OXS-1550 could deliver the most near-term opportunities. The company’s announcement that it would be working with a “major pharmaceutical company” may suggest that the company could be positioning itself for a strategic investment/partnership at the very least.
Though the major company was left nameless, we can infer from comments made by Dr. Daniel Vallera that the “multi-billion dollar oncology drug” is ibrutinib. The statement made in a company press release specifically stated, “e are very excited about our progress with GT’s OXS-1550 (DT2219) combined with ibrutinib, a potent small molecule Bruton Tyrosine Kinase (BTK) inhibitor which is already an established chemotherapeutic agent. We believe combination therapies like these that kill cancer cells based on entirely different mechanisms are the future of cancer treatment.”
Knowing this, we can begin connecting dots to the companies connected to ibrutinib. Known by its trade name, Imbruvica. This is a drug developed by Pharmacyclics and Janssen. Both Pharmacyclics and Janssen are subsidiaries of major pharmaceutical companies; Abbvie (ABBV) and Johnson & Johnson (JNJ) respectively.
Ibrutinib has already shown efficacy with other combination therapies like Venetoclax (marketed by Abbvie & Genentech) as well as Rituxan (rituximab), currently co-marketed by Biogen (BIIB) and Genentech in the U.S., for the treatment of patients with a rare type of non-Hodgkin’s lymphoma called Waldenström’s macroglobulinemia. The U.S. Food and Drug Administration recently approved Imbruvica (ibrutinib) plus Rituxan (rituximab) as a treatment.
GT’s products will need to continue to produce positive results in order for investors to remain interested.
Biopharmaceutical companies often get acquired or sell their patents after they have released early success in their clinical trials. Last year, Gilead Sciences Inc. acquired Kite Pharma Inc. in a deal that was valued at nearly $12 billion. This year we’ve already begun to see the acquisition trend grow after Bioverativ Inc. (BIVV) was acquired by French biotech company, Sanofi SA ADR in an $11.6 billion deal. Furthermore, Celgene Corp. (CELG) has recently acquired Juno Therapeutics Inc., which valued Juno at about $9 billion in the transaction. Novartis made a strategic deal in April with its $8.7 billion buyout of AveXis.
Though the near-term options for a full acquisition may not be realistic at this stage, it wouldn’t be out of the question for GT Biopharma to obtain a strategic investment similar to the Affimed/Genentech deal in our opinion.
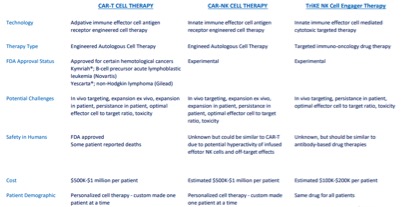
Both platforms utilize fusion proteins with one end binding to NK cells and the other targeting a tumor antigen, however, there are still a number of differences in the platforms. Many of these differences are based on exactly how the proteins are created and how they face the NK activation and proliferation issues.
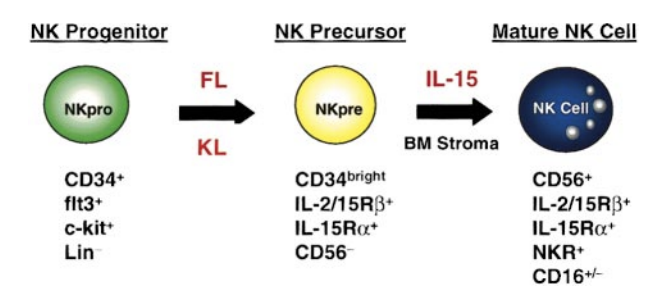
Essentially, without a specific stimulatory agent, the cells become exhausted as their numbers decline. Specifically, GT Biopharma’s NK cell-engager platform incorporates Interleukin (IL)-15. No other, including Affimed’s, NK cell technology has this.
From other research done on NK cells, it has been found that boosting NK cell numbers by treating them with IL-15 may be a valuable new approach to boosting our immunity to viral infections or cancer. IL-15 is a growth factor for certain kinds of white blood cells including natural killer cells and T cells.
GT Biopharma’s TriKE (Tri-specific Killer Engager) technology has shown superior NK cell cytotoxicity against hematologic tumors in preclinical work compared to earlier versions of the drug. TriKEs are an off the shelf option that employs a single chain, tri-specific fusion protein that binds to CD16 on NK cells and targets tumor antigens on cancer cells such CD33 in myeloid malignancies.
TriKEs are particularly attractive due to their low cost of manufacture relative to CAR-T therapy and can be used off the shelf without any modifications of autologous cells in the lab. The company is currently in the process of submitting an investigational new drug application to the FDA for the TriKE in order to launch a Phase I/II trial in the second half of this year.
This, in addition to several other key therapies in the company’s pipeline, could bode as attractive targets for larger investment from the likes of major pharmaceutical companies. This next combination trial will also be a proving ground for GT Biopharma to continue its progress on the early successes that OXS-1550 has already achieved.
Further Observation Is Needed
Right now researchers are looking at harnessing so-called “natural killer” cells to attack cancer in a way that may be more effective than other immunotherapies. Where CAR-T developed the pathway to new options for immunotherapy, NK cells may pave the way to new options with greater efficacy and affordability to address a number of cancers, today.
GT Biopharma has hit a number of milestones during its early phase trials. More observation is needed to determine the true potential of the company and its pipeline of treatments. The fact that a number of large pharmaceutical companies are beginning to heavily invest in this treatment option could be a sign that a new wave is building in biopharma development & immunotherapies in general. The Roche/Genetech deal may be the first of many geared toward NK cell advancement.
GT Biopharma is developing its NK enabling platform and bispecific ADC to address a variety of indications in myeloid malignancies and B-cell leukemias and lymphomas. There are roughly 30,000 CD33+ myeloid malignancy cases in the United States per year*.
B-cell leukemia and lymphoma cases are approximately 100,000 per year*. While there are other first-line therapies that could place these cancers in remission, many will not respond favorably and will require further treatment if the patient body allows it. Look at some of the more established treatments that we know about today like Merck’s Keytruda, which was granted initial approval for second-line treatment in patients with advanced or unresectable disease who no longer responded to other drugs.
Keytruda was better understood over time and as a result, it moved to first-line treatment in certain cases. While GT Biopharma may not see this right around the corner during the introduction of commercial-scale NK treatment, there is a criterion for immunotherapies to take the place of other approaches that may have more adverse side effects.
GT Biopharma’s positioning with a major pharmaceutical company to run combination trials with a proven drug in ibrutinib could result in future opportunities for the company. Considering that this is just one of many promising treatments in GT Biopharma’s pipeline, core management experience could warrant more attention from the investment community once preliminary results are reported from these initial combination trials.
Footnotes:
*American Cancer Society: Cancer Facts and Figures 2018
Disclaimer: Pursuant to an agreement between an affiliate of MAPH Enterprises, LLC, Midam Ventures LLC and GT Biopharma, Midam has been paid $200,000 from the company and ZERO shares of GT Biopharma Inc. for a duration beginning July 16, 2018 and ending August 16, 2018. Midam Ventures has been paid an additional $200k by GT Biopharma and has extended its marketing period through Sept 16, 2018. We may buy or sell additional shares of (GTBP) in the open market at any time, including before, during or after the Website and Information, provide public dissemination of favorable Information.
MAPH Enterprises, LLC | (305) 414-0128 | 1501 Venera Ave, Coral Gables, FL 33146 | new@marijuanastocks.com